>
>
>
The Multiplex Assay, Part 1: Where are all the Multiplexed LFIs?
The Multiplex Assay, Part 1: Where are all the Multiplexed LFIs?
- By Brendan O’Farrell, Ph.D.
- June 6, 2017
At DCN Dx, the demand for developing multiplex lateral flow devices is on the rise. Multiplex assays, which detect or measure multiple analytes in a single device, are a growing area of application development. However, designing, developing, and commercializing these products involve specific considerations.
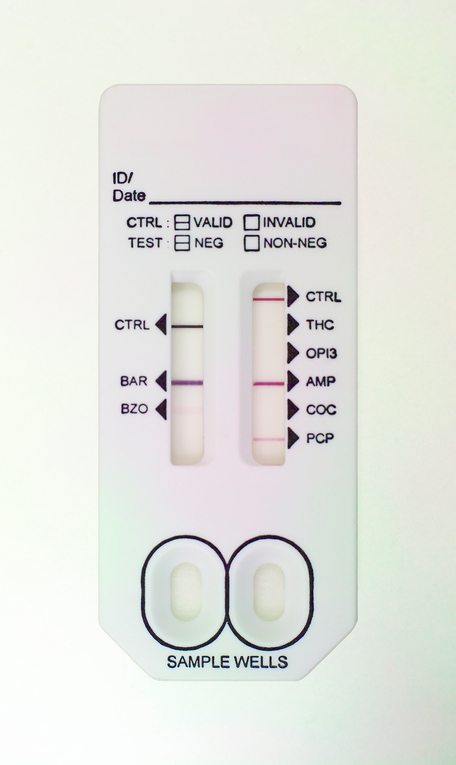
The term “multiplexing” can have different meanings, depending on the context. In general, a “multiplexed” assay detects or measures more than one analyte in a single device. However, it can also refer to generating multiple replicates of a single test within a single device or using positive, negative, or kinetic controls within a test, along with the analyte-specific assay.
Despite the interest in multiplex assays, the number of true multiplex lateral flow applications commercialized to date remains relatively low. This is due to various technical, manufacturing, and commercial considerations. In many cases, technical and manufacturing challenges are less of an obstacle than commercial ones. In this article, we will discuss these factors impacting the success of multiplexed devices.
Technical Issues
Multiplexing analytes is easier for certain classes of assays, such as serology tests, which target a single class of antibody directed against multiple analytes. In contrast, biomarker assays, which detect several different proteins from a single sample and may require multiple conjugates, are more complex.
Let’s take a closer look at a simple duplex system designed to detect two protein antigens in a whole blood sample. The primary technical considerations include:
- the specificity of the available binding reagents
- potential interferences in the sample
- the availability of the analytes for detection. In other words, does the sample have to be pre-treated and does the same sample treatment work for both analytes?
- the required LOD and dynamic range for each analyte. Are they present in roughly the same concentrations so that both can be detected from the same sample?
- the stability of the binding reagents for each analyte. Is any difference in degradation rates of those binding reagents?
Compatibility between the assays is crucial for multiplexing analytes from a single sample. If that compatibility exists, we can move on to the lower order technical hurdles. Not all of these issues are deal-killers. Many can be overcome. However, the more analytes we add to the mix, the greater the complexity of answering some of these questions becomes. If we add quantification to the mix, the system becomes even more complex. In that case, we are concerned with variability in the system that can limit the accuracy of the results.
We have to work to generate highly reproducible results in each of our assays and understand the issues around degradation of reagent performance in the context of what that can do to the standard curves in the system. Again, more analytes, more complexity.
Manufacturing Issues
Manufacturing a multiplexed lateral flow assay can be complex. The more assays on a device, the greater the potential risk of manufacturing failure and post-market failure of the product. Processing equipment, materials, manufacturing, and QC processes may differ from those used in standard single-plex assays, requiring manufacturers to have deep technical capabilities to ensure ongoing quality and production yields.
Commercial Issues
From a technical perspective, it may be possible to multiplex many different assays onto a single rapid assay product. However, the utility and market demand for the product must be considered. Questions to ask include:
- What is the added regulatory complexity of the multiplex relative to single-plexed assays?
- What is the added manufacturing complexity and risk?
- What is the likely cost of the device?
- Can we get reimbursement for the multiplexed assays?
- Does the market, the clinician, or the end user want or need all the data from our multiplex system?
Although there is a relatively small number of panels that make sense from all these perspectives, many assays can benefit from the power of replicates and built-in controls offered by a multiplexed assay.
So, why aren’t there more multiplexed lateral flow tests on the market? In short the ability to develop, manufacture and commercialize a multiplexed point of care test requires a balance between technical capability, design and market need. But there is demand in many application areas that can help to overcome the commercial issues, so developers and manufacturers need to be ready to meet the technical challenges. In part 2 of this series, we will discuss some effective multiplexed assay and product architectures and some novel approaches to overcome those challenges.
In the meantime, do not hesitate to contact us to discuss this topic further. Learn more about our lateral flow assay development services and our lateral flow education courses.
We also invite you to join us at the Advanced Lateral Flow Conference in San Diego, where lateral flow professionals from all over the world are gathering to learn, collaborate, and share ideas about lateral flow technology and the future of IVD the market. Learn more here.
Latest News
March 18, 2024